Expanding Availability of TTFields for Mesothelioma Patients
Tumor Treating Fields (TTFields) are a newer treatment option in the mesothelioma community. TTFields target mesothelioma cancer by producing low-intensity electrical currents. These disrupt cell division to slow or stop cancer growth.
In 2019, the United States Food and Drug Administration (FDA) first approved TTFields. Specifically, it approved use of the NovoTTF-100L™ System with pemetrexed and platinum-based chemotherapy. This treatment may be a first-line option for patients with pleural mesothelioma.
Request a Free 2020 Mesothelioma Guide
What Is the Significance of TTFields for Mesothelioma Patients?
TTFields can help improve mesothelioma survival. Recent studies demonstrated improved 1- and 2-year survival rates. Researchers also found success in shrinking tumors and stopping tumor growth.
Promising study results contributed to the FDA approval. Because there is no cure for mesothelioma, researchers look for treatments that will improve survival and decrease symptoms for patients. TTFields continue to demonstrate success in extending patient survival.
How Do TTFields Work for Mesothelioma?
TTFields target mesothelioma cell division. Low-intensity, wave-like electrical fields disrupt cancer cell division. Patients may experience tumors shrinking or a stop in tumor growth.
TTFields are combined with chemotherapy. Chemotherapy can also help stop cancer from spreading and kill remaining cancer cells.
What Does the Treatment Involve?
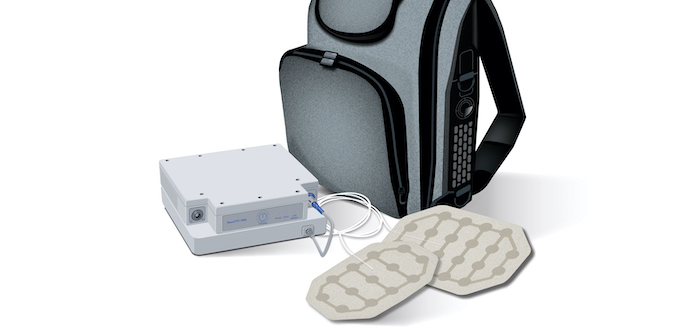
TTFields offer convenience to patients with a wearable, portable system. The device approved by the FDA is Optune Lua™ by Novocure®. The system:
- Is held within a wearable backpack
- Operates within the backpack for portable treatment
- May be used at night while the patient sleeps
- Allows patients to continue with many day-to-day activities
- Operates with rechargeable batteries
Patients should discuss logistics with their physician. There may be limitations specific to a patient’s individual case, particularly when undergoing chemotherapy treatment as well.
Side Effects of TTFields for Mesothelioma Patients
Researchers found limited side effects from TTFields. In one clinical study, the only side effect was skin irritation. The Optune Lua™ website notes patients should notify their physician if skin problems emerge. Some skin conditions can be treated with topical ointments and creams.
Compared to other aggressive cancer treatments, TTFields often exhibit less negative effects. TTFields target the cancer location instead of spreading through the patient’s entire body. This helps mitigate side effects.
Where Can Mesothelioma Patients Find Treatment With TTFields?
The availability of TTFields is expanding across the United States. One of the most recent cancer centers to offer the treatment is Moffitt Cancer Institute. The Optune Lua™ website offers a full list of participating centers. This includes (but is not limited to):
- Banner MD Anderson Cancer Center (Arizona)
- University of California, Los Angeles (California)
- Moffitt Cancer Institute (Florida)
- Brigham and Women’s Hospital (Massachusetts)
- Cleveland Clinic (Ohio)
- Baylor College of Medicine (Texas)
In total, more than 70 cancer centers are certified to offer this treatment option. The list continues to expand, making TTFields more accessible to mesothelioma patients.
Determining if TTFields Are the Best Treatment Option
Interested patients should ask their healthcare provider about TTFields. What’s best for one patient may not be best for another. Currently, Optune Lua™ is:
- For adult patients
- For patients with malignant pleural mesothelioma (MPM)
- For those with unresectable, locally advanced or metastatic MPM
- To be used with standard chemotherapy (pemetrexed and platinum-based chemotherapy)
A mesothelioma specialist can provide patients with a complete understanding of eligibility. If ineligible, there are other mesothelioma treatments that may be an option. Researchers continue to develop treatment advancements to help improve disease management for patients.